Specification
- ಔಷಧದ ಮೂಲ
- India
- ಸೂಚನೆ
- Neovascular (wet) age-related macular degeneration, Diabetic macular edema, Retinal vein occlusion, Myopic choroidal neovascularization
- ಉಪ್ಪು ಸಂಯೋಜನೆ
- Ranibizumab 2.3mg
- ಡೋಸೇಜ್ ಫಾರ್ಮ್
- Injection
- ಪ್ಯಾಕೇಜಿಂಗ್ (ಪ್ರತಿ ಪೆಟ್ಟಿಗೆಗೆ ಪ್ರಮಾಣ)
- Single vial per box
- ಡ್ರಗ್ ವಿಧ
- ಪದಾರ್ಥಗಳು
- Ranibizumab
- ಶಾರೀರಿಕ ರೂಪ
- ಕಾರ್ಯ
- ಇದಕ್ಕಾಗಿ ಶಿಫಾರಸು ಮಾಡಲಾಗಿದೆ
- Adults with neovascular (wet) age-related macular degeneration and other retinal diseases
- ಡೋಸೇಜ್
- 2.3mg per vial
- ಡೋಸೇಜ್ ಮಾರ್ಗಸೂಚಿಗಳು
- As prescribed by physician; for intravitreal use only
- ಸೂಕ್ತವಾಗಿದೆ
- ಪ್ರಮಾಣ
- 1 Vial
- ಶೇಖರಣಾ ಸೂಚನೆಗಳು
- Store at 2°C to 8°C. Do not freeze.
- Prescription Required
- Yes
- Container Type
- Glass Vial
- Appearance
- Clear to slightly opalescent, colourless to pale yellow liquid
- Therapeutic Class
- Ophthalmic drugs (anti-VEGF)
- Route of Administration
- Intravitreal
- Shelf Life
- 24 months from the date of manufacture
- Marketed By
- Intas Pharmaceuticals Ltd.
- Medicine Form
- Sterile solution for Injection
About
Razumab 2.3mg Injection is used for the treatment of eye diseases which are caused by diabetes, macular degeneration, and macular swelling. It reduces the abnormal growth and leakage of blood vessels in the eye that can cause vision loss.Razumab 2.3mg Injection is administered directly into the eyeball by an eye specialist. You should not self administer this medicine at home. The common side effects include conjunctival hemorrhage, eye pain, eye floaters, increased intraocular pressure, and eye inflammation.However, if you experience these side effects for a longer duration or any other symptoms which you think may be due to this medicine, please inform your doctor. It is not advised to drive after administration of this injection as it may cause blurring of vision.How Does Razumab Work?
Razumab (Ranibizumab 2.3mg) is an anti-VEGF (vascular endothelial growth factor) therapy that inhibits the action of VEGF, a protein responsible for abnormal blood vessel formation and leakage in the eye. By blocking VEGF, Razumab helps prevent further vision loss and may even improve vision in retinal diseases.
When Is Razumab Recommended?
Razumab is indicated primarily for adults diagnosed with neovascular (wet) age-related macular degeneration, diabetic macular edema, retinal vein occlusion, or myopic choroidal neovascularization. It is typically prescribed following a thorough ophthalmic evaluation to determine eligibility and expected benefit.
Proper Storage and Handling of Razumab
Store Razumab injection vials in a refrigerator at a temperature between 2C to 8C. Avoid freezing and keep the vial protected from light. Use the sterile solution directly from the single-use glass vial and discard any unused portion. Handle the injection under aseptic conditions as recommended for all intravitreal medications.
FAQ's of Razumab 2.3mg Injection:
Q: How is Razumab 2.3mg Injection administered?
A: Razumab 2.3mg Injection is administered directly into the eye (intravitreal injection) by a trained ophthalmologist in a clinical setting. It is not for self-administration and must be given under sterile conditions to minimize the risk of infection.Q: What eye conditions are treated with Razumab?
A: Razumab 2.3mg Injection is used to treat adults with neovascular (wet) age-related macular degeneration, diabetic macular edema, retinal vein occlusion, and myopic choroidal neovascularization. These conditions involve abnormal blood vessel growth or leakage within the retina.Q: When should I expect to notice improvement after receiving Razumab?
A: Response to Razumab may vary by individual and condition. Some patients notice improvement in vision within the first few injections, while others may need longer for noticeable changes. Regular follow-up appointments with your eye specialist are essential to monitor progress.Q: Where should Razumab 2.3mg Injection be stored before use?
A: Keep Razumab stored in its original package within a refrigerator at 2C to 8C, without freezing. Do not use the solution if it has been exposed to extremes of temperature or appears cloudy or discolored beyond pale yellow.Q: What is the process for using Razumab 2.3mg Injection?
A: The injection is given by an ophthalmologist after numbing your eye and cleaning the area. The entire process, including monitoring afterwards, typically takes less than an hour in a clinic setting. The dose and frequency are defined by your physician based on your specific eye condition.Q: What are the benefits of using Razumab for retinal diseases?
A: The primary benefit of Razumab is its ability to slow or stop vision loss caused by abnormal blood vessel growth or leakage in the retina. For some patients, Razumab can also help recover some lost vision, improving quality of life and daily functioning.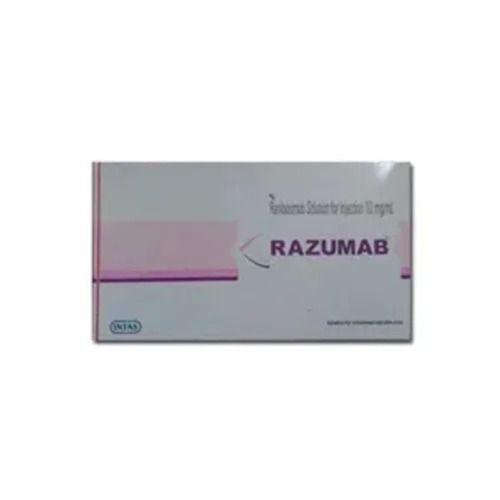
Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
ಮೊಬೈಲ್ number
Email
ಇನ್ನಷ್ಟು Products in ಫಾರ್ಮಾಸ್ಯುಟಿಕಲ್ ಇಂಜೆಕ್ಷನ್ Category
ಶೇಖರಣಾ ಸೂಚನೆಗಳು : Store below 25°C
ಕನಿಷ್ಠ ಆದೇಶ ಪ್ರಮಾಣ : 10
ಅಳತೆಯ ಘಟಕ : Vial/Vials
ಶಾರೀರಿಕ ರೂಪ : Liquid
ಡೋಸೇಜ್ ಮಾರ್ಗಸೂಚಿಗಳು : Bacterial infections, hospitalacquired and ventilatorassociated pneumonia, urinary tract infections, intraabdominal infections, skin and soft tissue infections, and uterine infections.
ಡೋಸೇಜ್ : 600
ಜಿವಿಬಾಕ್ 2.5 ಗ್ರಾಂ
ಶೇಖರಣಾ ಸೂಚನೆಗಳು : ಡ್ರೈ ಪ್ಲೇಸ್
ಕನಿಷ್ಠ ಆದೇಶ ಪ್ರಮಾಣ : 10
ಅಳತೆಯ ಘಟಕ : ಬಾಟಲುಗಳು/ಬಾಟಲುಗಳು
ಶಾರೀರಿಕ ರೂಪ : ದ್ರವ
ಡೋಸೇಜ್ ಮಾರ್ಗಸೂಚಿಗಳು : ಅಗತ್ಯವಿರುವಂತೆ
ಡೋಸೇಜ್ : ಸೂಚನೆಯ ಪ್ರಕಾರ
ಸೂರ್ಯ ಸ್ಪೆಷಾಲಿಟಿ ಫಾರ್ಮಾ
GST : 29AXZPT5360J1ZC
GST : 29AXZPT5360J1ZC
Bno No - 33, ನೆಲ ಮಹಡಿ, 14ನೇ ಮುಖ್ಯ ಕುಮಾರಸ್ವಾಮಿ ಲೇಔಟ್, 1 ಸ್ಟ ಹಂತ,ಬೆಂಗಳೂರು - 560078, ಕರ್ನಾಟಕ, ಭಾರತ
ದೂರವಾಣಿ :08045804285
|
 |
SURYA SPECIALITY PHARMA
ಎಲ್ಲ ಹಕ್ಕುಗಳನ್ನು ಕಾಯ್ದಿರಿಸಲಾಗಿದೆ.(ಬಳಕೆಯ ನಿಯಮಗಳು) ಇನ್ಫೋಕಾಮ್ ನೆಟ್ವರ್ಕ್ ಪ್ರೈವೇಟ್ ಲಿಮಿಟೆಡ್ . ಇವರಿಂದ ಅಭಿವೃದ್ಧಿಪಡಿಸಲಾಗಿದೆ ಮತ್ತು ನಿರ್ವಹಿಸಲಾಗಿದೆ |

 ವಿಚಾರಣೆಯನ್ನು ಕಳುಹಿಸಿ
ವಿಚಾರಣೆಯನ್ನು ಕಳುಹಿಸಿ